Fast Track guides you step by step to a CE-ready submission — no guesswork, just clarity.
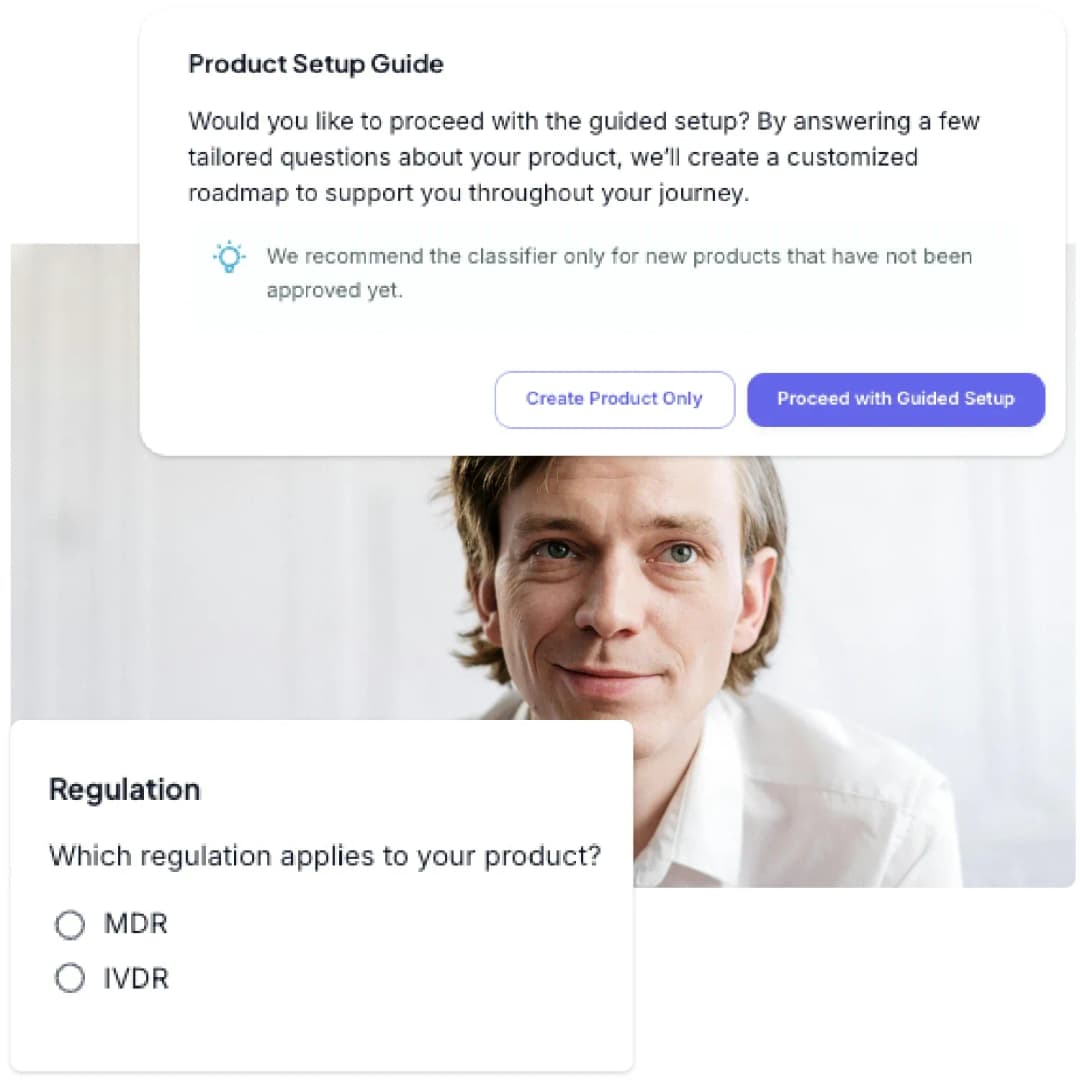
From zero to hero in weeks: Go from no documentation to a full CE submission package faster than ever.
Guided onboarding: Our superb customer success guides you step by step through what you have to do, in person and with on-demand tutorials.
Submission-ready documentation: Automatically generated, fully traceable, and aligned with regulatory expectations.
Expert-reviewed logic: Templates and decision flows are backed by regulatory experts for peace of mind.
Fraction of costs: Achieve higher outcome with fewer costs than every before thanks to newest technologies and the right experts jumping if when really needed.

From zero to hero in weeks: Go from no documentation to a full CE submission package faster than ever.
Guided onboarding: Our superb customer success guides you step by step through what you have to do, in person and with on-demand tutorials.
Submission-ready documentation: Automatically generated, fully traceable, and aligned with regulatory expectations.
Expert-reviewed logic: Templates and decision flows are backed by regulatory experts for peace of mind.
Fraction of costs: Achieve higher outcome with fewer costs than every before thanks to newest technologies and the right experts jumping if when really needed.
From the first approval to every update and audit, CertHub ensures your compliance evolves with your product.

Generate TechDocs instantly: Create full MDR/IVDR submissions with one click — from Risk Files to Clinical Evaluations.
Start from scratch: Use our Knowledge Model for new products or let AI convert Word/Excel chaos into structured documentation.
Edit once, update everywhere: Change a single element, and updates flow automatically into every related document and submission.
Full traceability built in: Every paragraph links back to its source, ensuring transparency and audit confidence.
Always know what’s missing: Dashboards show completeness, gaps, and quality signals — so you submit only when ready.
Spot gaps early: Run an AI-powered gap analysis to identify missing or weak documentation before you invest time in writing.
Track progress live: Monitor what’s complete, missing, or outdated across all submission modules with real-time dashboards.
Simulate audits: Test your QMS against an AI-driven audit checker that highlights structural and semantic issues before the real thing.
Check submissions before handoff: Upload your TechDoc package and get a final AI review against Notified Body expectations, reducing the risk of rejection.
De-risk certification: Fix problems before they cause delays, costly rework, or failed submissions — and enter your next audit with confidence.


Multi-standard coverag: Checks conformity against ISO 13485, ISO 13495, ISO 14971, IEC 62366, and EN ISO 15189 — all in a single workflow.
Root-cause diagnosis: Pinpoints gaps and non-conformities in your QMS documentation before an external auditor does, so your team can act early.
Four-stage audit simulation: Walks you through scope definition, document upload, simulated audit, and a full remediation plan with risk analysis.
Audit-ready reports: Generates structured findings, corrective actions, and risk assessments ready to present to certification bodies.
Cut prep time by up to 57 h: What normally takes weeks of manual review can now be completed in a fraction of the time, letting your team focus on quality, not paperwork.

Generate TechDocs instantly: Create full MDR/IVDR submissions with one click — from Risk Files to Clinical Evaluations.
Start from scratch: Use our Knowledge Model for new products or let AI convert Word/Excel chaos into structured documentation.
Edit once, update everywhere: Change a single element, and updates flow automatically into every related document and submission.
Full traceability built in: Every paragraph links back to its source, ensuring transparency and audit confidence.
Always know what’s missing: Dashboards show completeness, gaps, and quality signals — so you submit only when ready.

Spot gaps early: Run an AI-powered gap analysis to identify missing or weak documentation before you invest time in writing.
Track progress live: Monitor what’s complete, missing, or outdated across all submission modules with real-time dashboards.
Simulate audits: Test your QMS against an AI-driven audit checker that highlights structural and semantic issues before the real thing.
Check submissions before handoff: Upload your TechDoc package and get a final AI review against Notified Body expectations, reducing the risk of rejection.
De-risk certification: Fix problems before they cause delays, costly rework, or failed submissions — and enter your next audit with confidence.

Multi-standard coverag: Checks conformity against ISO 13485, ISO 13495, ISO 14971, IEC 62366, and EN ISO 15189 — all in a single workflow.
Root-cause diagnosis: Pinpoints gaps and non-conformities in your QMS documentation before an external auditor does, so your team can act early.
Four-stage audit simulation: Walks you through scope definition, document upload, simulated audit, and a full remediation plan with risk analysis.
Audit-ready reports: Generates structured findings, corrective actions, and risk assessments ready to present to certification bodies.
Cut prep time by up to 57 h: What normally takes weeks of manual review can now be completed in a fraction of the time, letting your team focus on quality, not paperwork.